
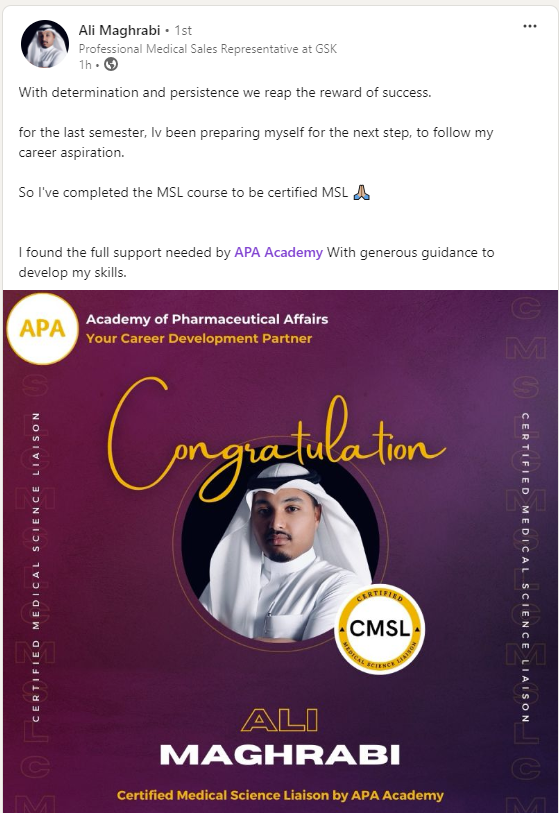
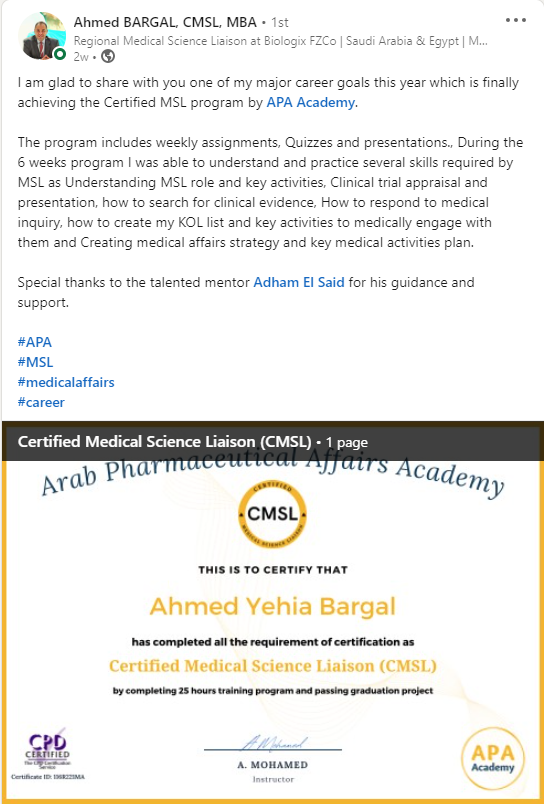
Build your transition to MSL role
Certified. Confident. Ready for Medical Affairs
Cohort 30
Starting April 17th, 2026
Designed for Product Specialists, Medical Representatives and Aspiring MSLs in the Middle East
CPD United Kingdom Accredited | 10 Online Live Sessions | MSL Assessment Support
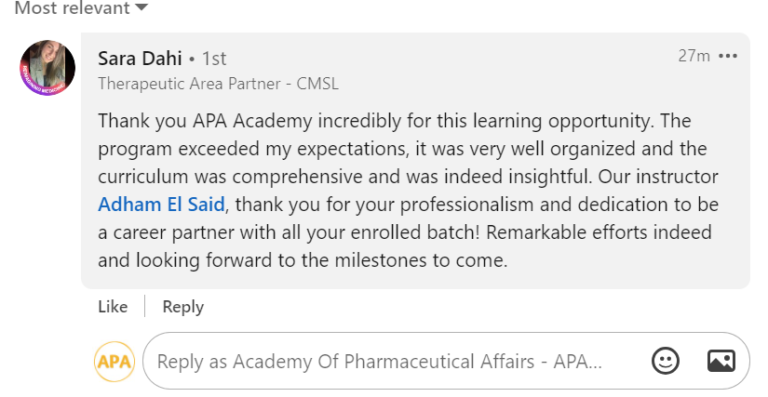
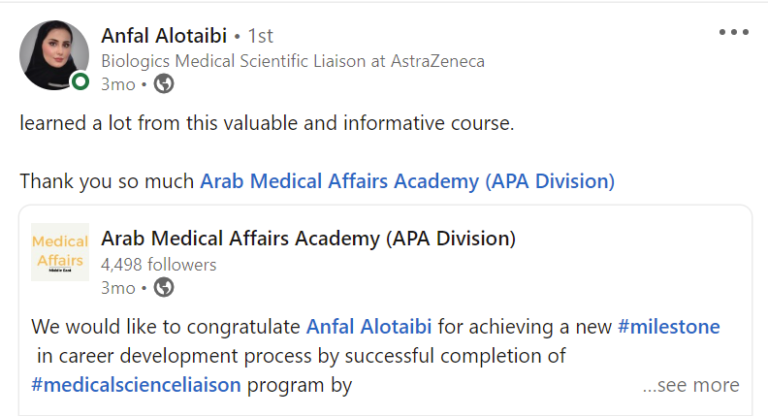
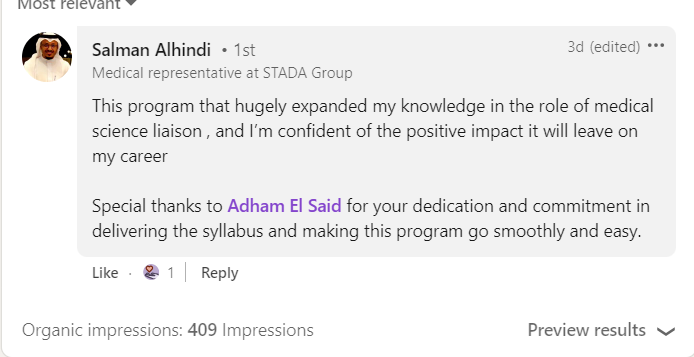
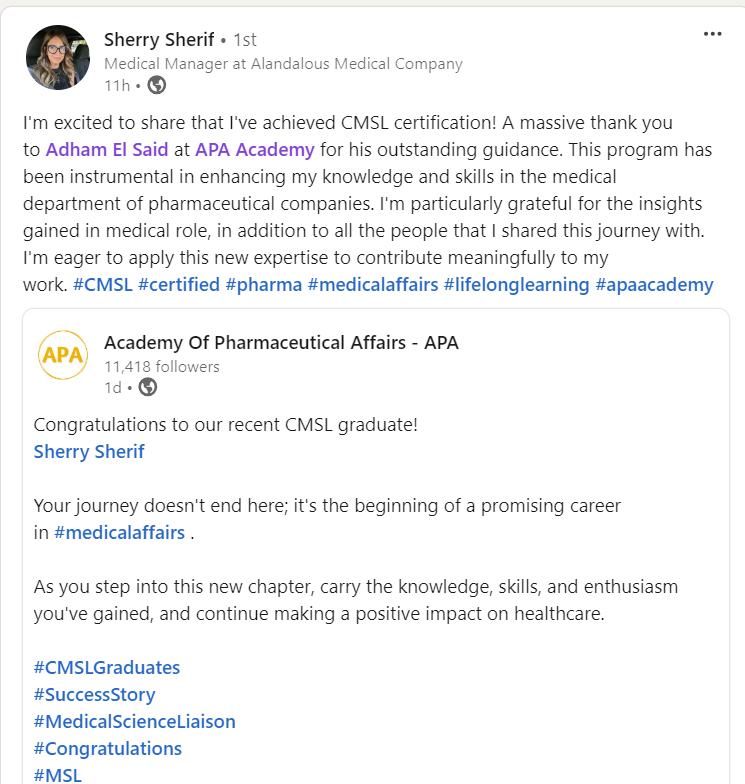
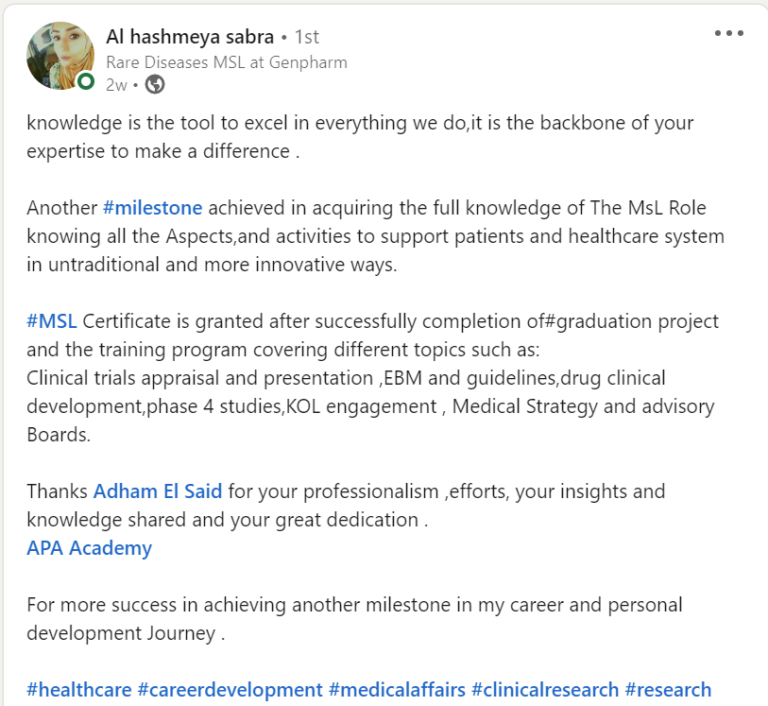
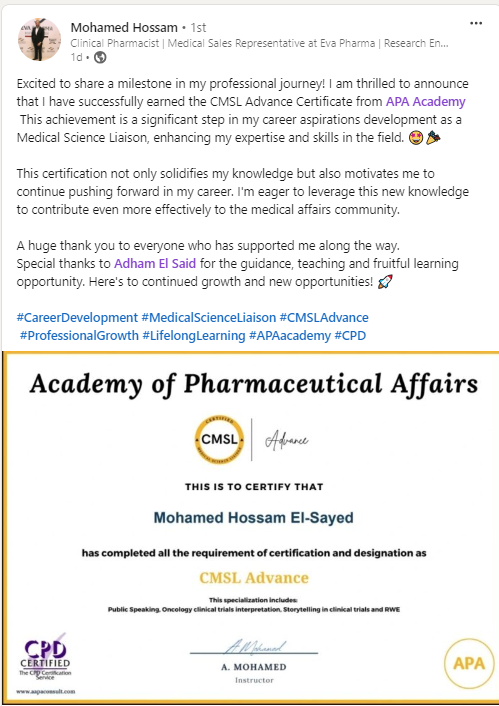
Join a community of more than 600 Graduate in the MENA Region in a Comprehensive Learning Journey Towards Medical Affairs





























Start Your Learning Journey Now
Learn. Apply. Lead.
The CMSL Certificate equips you with the MSL competencies, practical skills, and strategic insights you need to be ready for MSL Assessment.
10 Interactive Live Sessions
Master MSL Role, clinical trial appraisal and presentation, medical strategy development, KOL engagement, and effective medical communication.
3 Individual MSL Assessments
Receive personalized coaching, comprehensive assessments, and tailored guidance that extends beyond certification, ensuring impactful career development.
1 - Year Mentorship access
Enhance your professional growth with Quarterly Q & A sessions on different case studies and 5 sessions on business communication, Storytelling and interviewing skills, specifically designed to prepare you for successful MSL assessments.
CMSL Learning Objectives
By the end of the CMSL Certificate, you’ll be able to demonstrate technical, strategic, and communication competencies essential to succeed as a Medical Science Liaison.
Understand the MSL Role
- Define the role of an MSL and its significance in pharma.
- Identify the essential competencies and skills needed to be an effective MSL.
- Analyze the responsibilities and expectations of an MSL in different contexts.
Answer MSL Interview Questions
- Recognize typical MSL interview questions.
- Apply strategies to answer convincingly.
- Refine responses based on feedback.
Critically Appraise Trials
- Analyze trial design and components.
- Evaluate trial quality and validity.
- Present critical appraisals effectively.
Medical Strategy & Activities
- Develop comprehensive medical strategies.
- Execute activities across product lifecycle.
- Justify activity selection by stage/objectives.
KOL List & Engagement
- Define criteria for selecting KOLs.
- Build a therapeutic-area specific list.
- Develop strategies to engage and cultivate relationships with KOLs effectively.
Understand Clinical Terms
- Define key clinical trial terminologies.
- Apply them to interpret protocols/results.
- Communicate them clearly in practice.
Present Clinical Trials
- Deliver clear, engaging presentations.
- Organize and structure clinical content.
- Use visual aids effectively.
Guidelines & Medical Questions
- Interpret and apply medical guidelines.
- Formulate evidence-based responses.
- Address complex inquiries with clarity.
Understand PMOS Studies
- Differentiate post-marketing study types.
- Define their objectives and outcomes.
- Evaluate relevance to Medical Affairs.
Manage Advisory Boards
- Plan and lead effective advisory boards.
- Overcome key engagement challenges.
- Evaluate board effectiveness.
Risk & Safety Communication
- Explain MSL role in safety education.
- Train HCPs on risk minimization.
- Communicate safety impact clearly.
Core Expertise & Achievements
- Medical Affairs Leadership (10 yrs): Drove cross-functional launch readiness and KOL engagement programs that expanded therapeutic access across diverse markets in the GCC and Levant regions.
- Medical Governance & Compliance: Implemented region-wide SOPs and training that aligned global standards with local regulations, ensuring audit-ready operations.
- Clinical Research & RWE: Guided post-marketing observational research, bridging evidence gaps and informing real-world practice.
Credentials
- Board Certified Oncology Pharmacist (BCOP)
- Professional Certified Trainer – American University in Cairo & Association for Talent Development (ATD)
- Scientific Research & Biostatistics Program – King Abdul-Aziz University
Training & Coaching Impact
-
- CMSL Certificate Program: Trained 600+ aspiring MSLs across the Middle East.
- CMAP Medical Affairs Diploma: Developed and delivered curricula for 100+ practicing MSLs.
- 1,300+ one-to-one coaching sessions for current and future MSLs, sharpening scientific storytelling, compliance, and stakeholder engagement skills.
- 5 years of workshop facilitation on launch excellence, insights generation, and medical strategy.
Join graduates from more than 50 company across MENA Region





